The video below explains the different routes that can be taken and how to choose the best method. In order to calculate compensation, there are many options available: automated tools, manual adjustment, software on the cytometer, analysis software. More information on the best practices for creating compensation controls can be found in the Flow Basics 2.4 video, which discusses all experiment controls. Controls should have sufficient events in both positive and negative populations.The control should ideally contain a positive and negative population, and these populations must have the same autofluorescence properties. 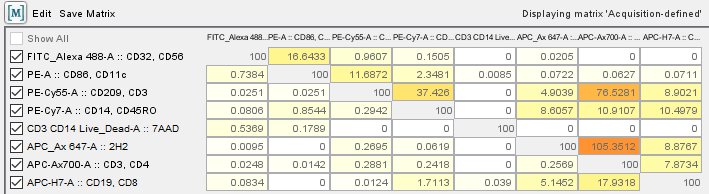
The control should ideally be exactly the same fluorophore as the one in the sample.The positive population must be as bright or brighter than the multicolor sample.There are several rules that must be followed to ensure that compensation can be properly calculated: However, if a small panel was designed in such a way that spillover between all flurophores is negligible then compensation is not needed. Proper compensation is necessary to ensure that false positives are not being analyzed. Bioinformatics, 2015 and CyTOF data PhenoGraph, Levine et al., Cell, 2015. This service is currently free of charge, too.Compensation is used to ensure that the signal in a single detector is coming from the assigned fluorophore and not spillover from another fluorophore in the panel. Seurat provides several useful ways of visualizing both cells and features that define the PCA, including VizDimReduction, DimPlot, and DimHeatmap pbmc <- RunPCA(pbmc) SaveObject(pbmc, ' seurat objafterPCA'). If you are sorting in the CFFC you will receive step-by-step tuition and guidance leading you to being able to work with the FACSAria sorter independently and operator-free. This workshop is co-organized by TransMed and the Core Facility Flow Cytometry (CFFC)Ībout the Core Facility Flow Cytometry (CFFC)Įveryone is welcome to use FlowJo TM on the CFFC workstations, currently free of charge. Please bring your own data with you in order to analyze them.įor the workshop 1 CP for scientific skills training can be credited (0.5 CP for participating only in the theoretical part). Click on the Record Background Gating Data option. Please read the FlowJo TM description, which will be sent to you after your registration. In order to calculate the background in FlowJo, you need to compensate your data in FlowJo. Theoretical and practical input by the trainer, showing examples, analyzing own data using FlowJo TM with the help of the trainer, discussing own issues, feedback from the trainer.ĭoctoral students, postdocs and clinician scientists who already know the basics of FlowJo TM. The steps shown in this tutorial will help you in the analysis of nearly any kind of FACS data. Using R-independent plugins with FlowJo TM FlowJo is capable of much that simply cant be covered in an introduction such as this (for example, there are Analysis Platforms to perform sophisticated DNA/Cell Cycle analysis, Kinetics analysis, Compensation, etc).Using R-dependent plugins with FlowJo TM.Editing an existing compensation matrix.Creating a compensation matrix using the Wizard.Heat mapping statistics in the Table and Layout Editors.Overlays - grouping by panel or keyword.General overview of FlowJo TM including the following:.There is enough time to discuss specific issues. In the afternoon, participants practice FlowJo in pairs using own data. Set the desired parameters for each axis and add required gates. Double-click on a file to open the plot window. Open FlowJo software, and drag and drop the Attune NxT data files to analyze in the software. FlowJo Software enables single-cell flow cytometry analysis that helps unlock your data insights with accessible features for immunophenotyping, cell cycle, proliferation, kinetics studies, quantitative population comparison, plate screening assays and more. In the morning, the software is explained theoretically using examples. instructions on defining a compensation matrix using compensation controls, refer to the user documentation for FlowJo software. FlowJo Software is your partner and committed to take your research to new heights. It was developed and incorporated into FlowJo v10.7 in collaboration with researchersfrom the Vlaams Instituut voor Biotechnologie in Belgium and the the Babraham Institute in Cambridge, UK. This beyond the basic workshop shows participants how to analyze flow cytometry data using FlowJo TM. AutoSpill is a new algorithm for calculating spillover and producing a compensation matrix. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
